Acetaminophen – Alternate USP Method
Acetaminophen and two of its major impurities were analyzed using a simple Mobile Phase. The Peak Shapes are very good and the Repeatability was good (%RSD = 0.01).
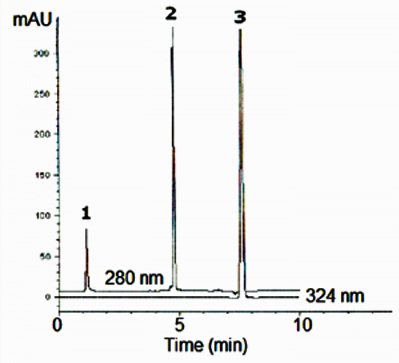
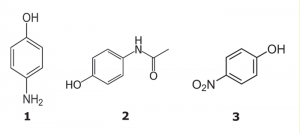 Peaks:
Peaks:
1. 4-Aminophenol 1.072 minutes
2. Acetaminophen 4.668 minutes
3. 4-nitrophenol 7.588 minutes
Method Conditions:
Column: Cogent Bidentate C18™, 4µm, 100Å
Catalog No.: 40018-75P
Dimensions: 4.6 x 75mm
Mobile Phase:
—A: DI Water with 0.1% Formic Acid
—B: Acetonitrile with 0.1% Formic Acid
Gradient:
| Time (minutes) | %B |
| 0 | 0 |
| 1 | 0 |
| 4 | 30 |
| 6 | 30 |
| 6.01 | 10 |
| 10 | 10 |
Post Time: 3 minutes
Injection Volume: 5µL
Flow Rate: 1.0mL / minute
Detection: UV
—280 nm (4-aminophenol, acetaminophen)
—324 nm (4-nitrophenol)
Sample Preparation: The stock solution was prepared by dissolving 1.0mg of standards in 10.00mL of the Mobile Phase (50:50 Solvent A / Solvent B). The Solution was then filtered with a 0.45µm Nylon Syringe Filter (MicroSolv Tech Corp.). The injection sample was diluted 1:10.
t0: 0.9 minutes
Note: Acetaminophen (n-acetyl-p- aminophenol, APAP) is a non-steroid anti- inflammatory drug. There are several impurities that can be present in a final drug (depending on the synthetic route, the quality of starting materials, reagents, etc.) which can have safety implications. Analytical methods are needed
to detect and identify these impurities and quantify them.
Attachment
No 249 Acetaminophen Impurities Method with HPLC pdf 0.3 Mb Download File


