Separation of Prodrug from Acid Hydrolysis Products
This Method shows the Separation of an ester Prodrug, Mometasone Furoate, from another compound formed during acid hydrolysis. This Peak is quite possibly the active form, Mometasone. The two Chromatograms show the non-degraded extract (Figure A) and an acid hydrolysis extract (Figure B).
Hydrolysis of the ester group would make the resulting Mometasone API less hydrophobic, and therefore earlier elution is predicted. Indeed, this is what is observed in the data. An excipient Peak from the cream matrix is also observed just after the solvent front and does not interfere with either the Prodrug peak or the API.
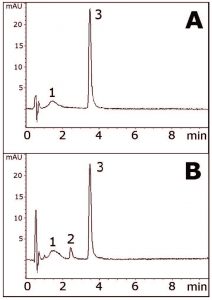
Peaks:
1. Excipient
2. Degradant (possibly Mometasone)
3. Mometasone Furoate (Prodrug)
Method Conditions
Column: Cogent Bidentate C8 2.o™, 2.2μm, 120Å
Catalog No.: 40208-05P-2
Dimensions: 2.1 x 50mm
Mobile Phase: 50:50 DI Water / Acetonitrile (v/v)
Injection vol.: 2μL
Flow rate: 0.3mL / minute
Detection: UV @ 248nm
Sample Preparation: 5.0g of a 0.1% Mometasone Furoate USP topical cream was weighed in a 250mL Erlenmeyer flask with a stirring bar. 50mL Methanol was added and the flask was stirred for 1 hour with a stopper covering the flask. Then a portion was filtered with a 0.45μm Nylon Syringe Filter (MicroSolv Tech Corp.). Next, one portion of the filtrate was diluted 1:5 with Methanol (Figure A) and a second portion was diluted 1:5 with a diluent of 90:10 Methanol / 1N HCL and heated in a dry bath at 80°C for 10 minutes. (Figure B).
Note: Mometasone Furoate is a prodrug where the API is formed by hydrolysis of the ester group. It is used to reduce inflammation due a variety of ailments and is available under trade names such as Elocon®, Novasone®, and Asmanex®. In addition to a topical ointment, the drug is also available as a dry powder inhaler or a nasal spray depending on the application.
Attachment
No 325 Mometasone Furoate in Topical Cream Analysis pdf 0.3 Mb Download File


