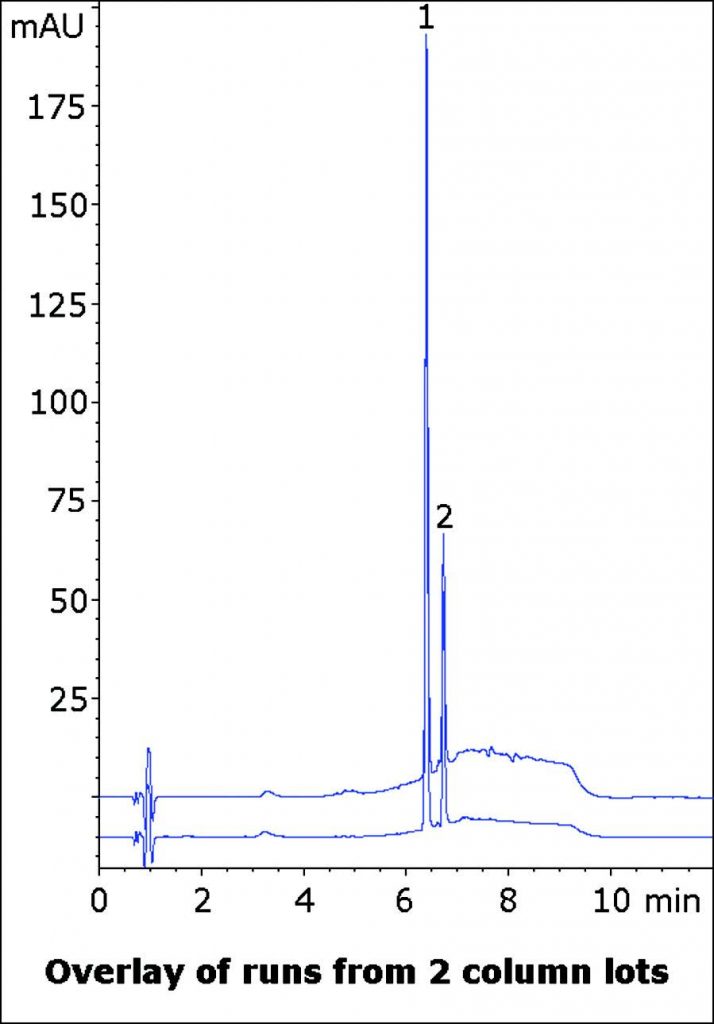
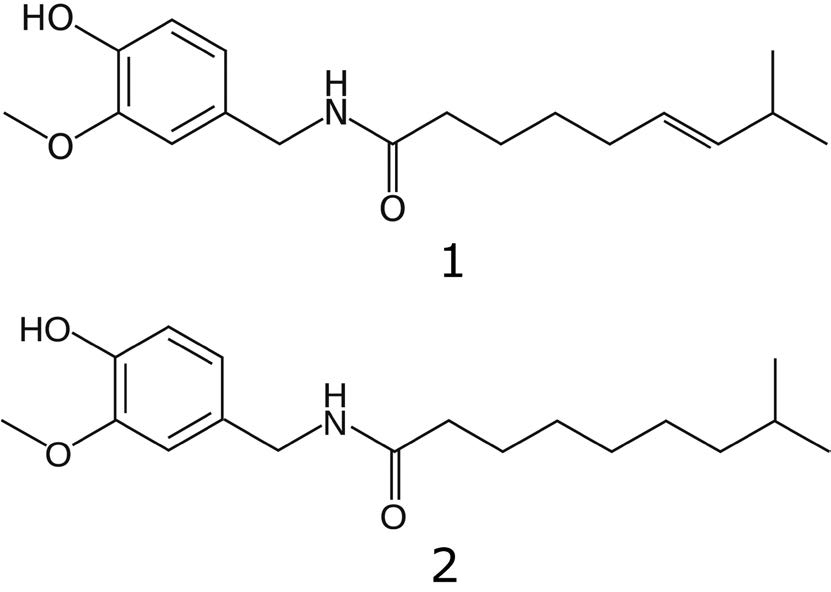
Separation of Capsaicinoids in an OTC Formulation
A formulation of Capsaicins is used as a topically applied solution for Arthritis pain relief. Analyzing Capsaicin and a similar compound, Dihydrocapsaicin is required. The Method illustrates the ability to resolve structurally similar Capsaicinoids from a pharmaceutical formulation. Peak Shapes and Efficiencies are excellent for both Peaks.
Furthermore, this AppNote presents data from two Column lots with the overlay of two runs in the figure below to exhibit Robustness and Precision of the Method.
Peaks:
1. Capsaicin
2. Dihydrocapsaicin
Method Conditions
Column: Cogent Bidentate C18™, 4μm, 100Å
Catalog No.: 40018-75P
Dimensions: 4.6 x 75mm
Mobile Phase:
—A: DI Water with 0.1% Formic Acid (v/v)
—B: Acetonitrile with 0.1% Formic Acid (v/v)
Gradient:
| Time (minutes) | %B |
| 0 | 20 |
| 1 | 20 |
| 3 | 80 |
| 8 | 80 |
| 9 | 20 |
Post Time: 3 minutes
Injection vol.: 1μL
Flow rate: 1.0mL / minute
Detection: UV @ 280nm
Sample Preparation: TopCare® Solution, a commercially available Pharmaceutical Product, containing 0.15% Capsaicin was filtered with a 0.45μm Nylon Syringe Filter (MicroSolv Tech Corp.). Peak identities were confirmed using individual USP Reference Standards.
t0: 0.9 minutes
Note: Capsaicin is the active ingredient in Chili Peppers and has many uses. It is commonly used in foods to add spiciness, but is also used in pepper spray. It is a highly potent irritant in mammals and pure Capsaicin reference standards should be handled with care.
Attachment
No 222 Capsaicin in Topical Solution Analyzed with HPLC pdf 0.3 Mb Download File