Method Optimization in Separation of Ethyl Glucuronide and Ethyl Sulfate
Ethyl Glucuronide and Ethyl Sulfate are compounds that are not well-retained or separated using Reversed Phase Columns due to their hydrophilicity thus requiring high water concentrations in the Mobile Phase.
To avoid the use of high water content Mobile Phases, HILIC columns were sought as a solution. Due to the mechanism of separation between the Mobile Phase and water-rich layer on the stationary phase, column equilibration was lengthy, noted to be around 14 minutes between run times. Cogent TYPE-C™ Silica was considered a good substitution for the mildly hydrophobic surface of the silica hydride material. The Cogent UDA™ column in conjunction with MS detection was shown to circumvent these issues.
The presented data illustrates the excellent separation and detection of these two compounds with rapid Column equilibration, being 2 minutes between gradient runs.
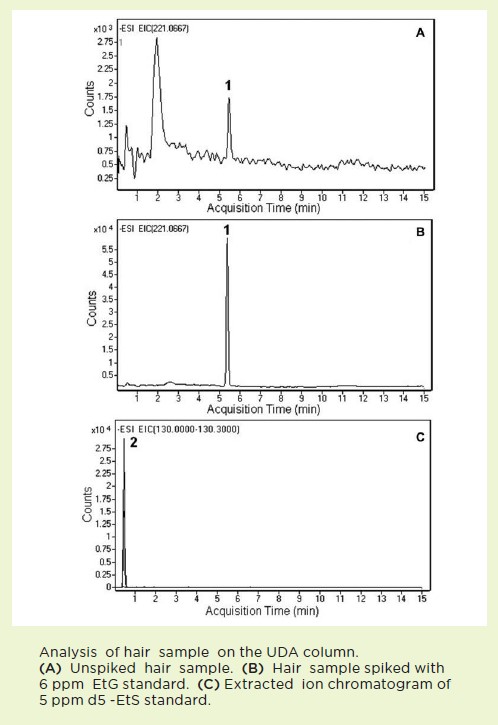
Peaks:
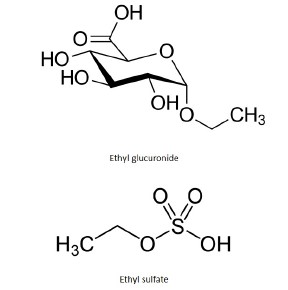
1. Ethyl Glucuronide – EtG, m/z = 221.0667
2. Ethyl Sulfate – EtS, m/z = 131.0295
Method Conditions:
Column: Cogent UDA™, 4µm, 100Å
Catalog No.: 40031-15P-2
Dimensions: 2.1 x 150 mm
Mobile Phase:
– A: DI Water/ 10 mM Ammonium Formate (v/v)
– B: 95% Acetonitrile/ 5% DI H2O/ 10 mM Ammonium Formate (v/v)
Gradient:
| Time (Minutes) | %B |
| 0 | 95 |
| 3 | 30 |
| 6 | 30 |
| 8 | 95 |
| 10 | 95 |
Flow rate: 0.4 mL/minute
Detection: ESI – NEG – Agilent 6220 MSD TOF Mass Spectrometer with a dual sprayer electrospray source
Sample: 0.2-0.7 mg/mL DI Water
Notes: Ethyl glucuronide and Ethyl Sulfate are metabolites of Ethanol, which are formed by enzymatic conjugation of Ethanol with Glucuronic Acid. This process, known as glucuronidation, is the result of exposure to Ethanol. These compounds are used as a long-term biomarkers in humans to test for alcohol abuse.
Attachment
Ethyl Glucuronide AppNote pdf 0.2 Mb Download File


