Temperature is a useful variable to consider in HPLC separations.
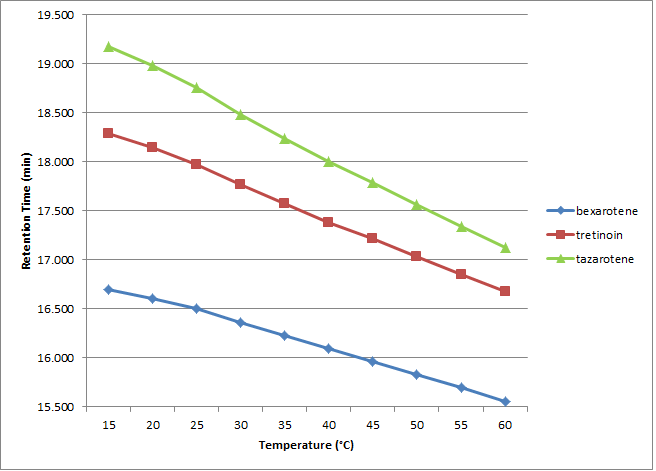
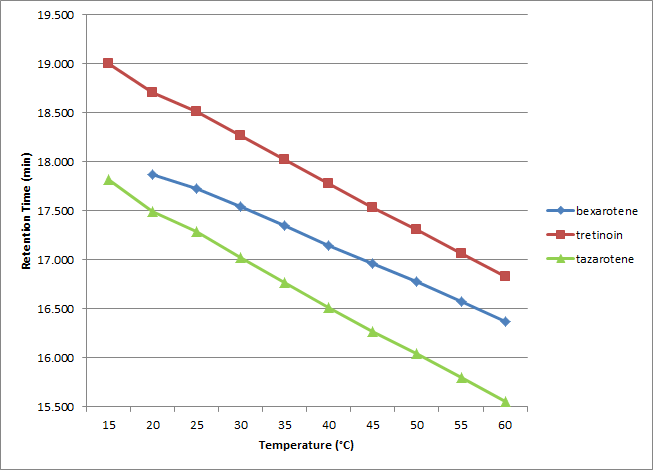
Control of temperature can reduce run times, alter selectivity, and change analyte efficiency. In this study of three terpenoids (bexarotene, tretinoin, and tazarotene), we compared the Cogent Bidentate C18™ and Cogent UDC-Cholesterol™ columns at different temperatures in an attempt to gain insight into their retentive behavior. The method conditions are shown in Table 1. Figures 1 and 2 show the retention times obtained for the three analytes as a function of temperature using each column. The UDC column is known to exhibit shape selectivity for some analytes, and we can see that the elution order is different when compared to the Bidentate C18.
The Bidentate C18 shows greater selectivity between the critical peak pair at higher temperature whereas the UDC shows the opposite trend. Since shape selectivity effects are known to be more pronounced when the UDC stationary phase moiety is more rigid, it makes sense that the lower temperatures would produce greater separation between the analytes. The Bidentate C18 stationary phase on the other hand does not have this property and therefore the effect is not the same. In fact, when using the Bidentate C18 at 15 °C where the UDC had shown the best separation, bexarotene appeared as a shoulder peak on the tazarotene peak.
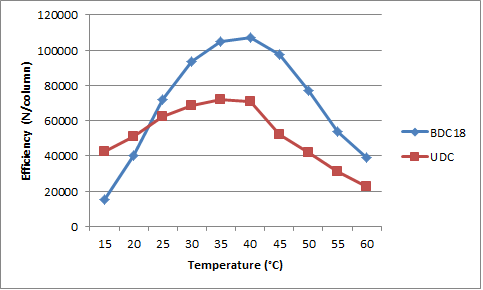
Peak efficiency initially increased with increasing temperature until it reached a maximum and then decreased (see Figure 3). The drop-off was more significant on the UDC column, suggesting that rigidity of the UDC moiety may play a more important role in the retention than the Bidentate C18. The maximum values were obtained in the range of 35–40 °C.
Knowing how temperature affects the separation can lead to more informed choices during method development. Resolution depends on both efficiency and selectivity, but their optimum values are not always the same; the selectivity on the UDC was best at 15 °C but the efficiency was highest at 35 °C. The analyst should consider the relative importance each effect that the temperature has on the chromatographic data when choosing an optimum column temperature.
Table 1. Method conditions for the separations.
| Parameter | Details |
|---|---|
| Solvent A | DI water + 0.1% formic acid |
| Solvent B | Acetonitrile + 0.1% formic acid |
| Flow Rate | 1.0 mL/min |
| Gradient | 0–1 min hold at 30%B, 1–24min to 100%B, 24–25min to 30%B |
| Detection | UV 254nm |
| Injection Volume | 10µL |