LC-MS Compatible HPLC Method for Tramadol
Tramadol can be retained by Reversed Phase HPLC but some reported methods require Phosphate buffer additives in the Mobile Phase, which means incompatibility with LC-MS. In contrast, this Method uses Formic Acid as the Mobile Phase additive.
An advantage this method has is that it produces enhanced signal-to-noise compared to Reversed Phase thereby producing better results. High Water content in the Reversed Phase Mobile Phase is often required for retention. Since Water is less volatile than Acetonitrile, it is more difficult to remove in LC-MS, hence also leading to lower sensitivity than this Method.
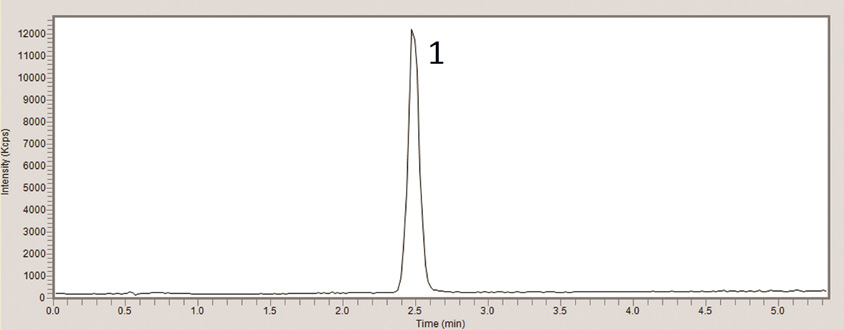
PEAK:
Tramadol, m/z 264.2 (M+H)+
Method Conditions
Column: Cogent Diamond Hydride™, 4μm, 100Å
Catalog No.: 70000-10P-2
Dimensions: 2.1 x 100mm
Mobile Phase:
—A: DI Water/ 0.1% Formic Acid
—B: Acetonitrile/ 0.1% Formic Acid
Gradient:
| Time (Minutes) | %B |
| 0 | 90 |
| 5 | 90 |
| 7 | 30 |
| 8 | 30 |
| 10 | 90 |
Injection vol.: 1μL
Flow rate: 0.4 mL/minute
Detection: ESI – POS – Agilent 6210 MSD TOF Mass Spectrometer
Sample Preparation: Tramadol HCl (50 mg) pill was ground with mortar and pestle into fine powder and was used to prepare 1 mg/mL stock solution in Milli-Q DI Water diluent. Then, the sample was sonicated for approximately 5 minutes prior to 0.45 um syringe filtration.
Note: Tramadol is an analgesic used to treat moderate to moderately severe pain. It can be used in both human and veterinary applications. It is sold under various formulation types and brand names, including Ryzolt®, Ultracet®, and Ultram®.
Attachment
No 363 Tramadol.pdf 0.2 Mb Download File


