Comparing cetylpyridinium chloride adsorption in glass autosampler vials
This study investigated the effect of analyte loss due to adsorption by hydroxyl groups (aka silanols) on the glass surface of autosampler vials over time. It is well known that the surface of standard, including industry leading borosilicate glass vials are populated with silanols and basic analytes can bind with these sites electrostatically or via hydrogen-bonding, leading to lower sample recovery in HPLC and LCMS.
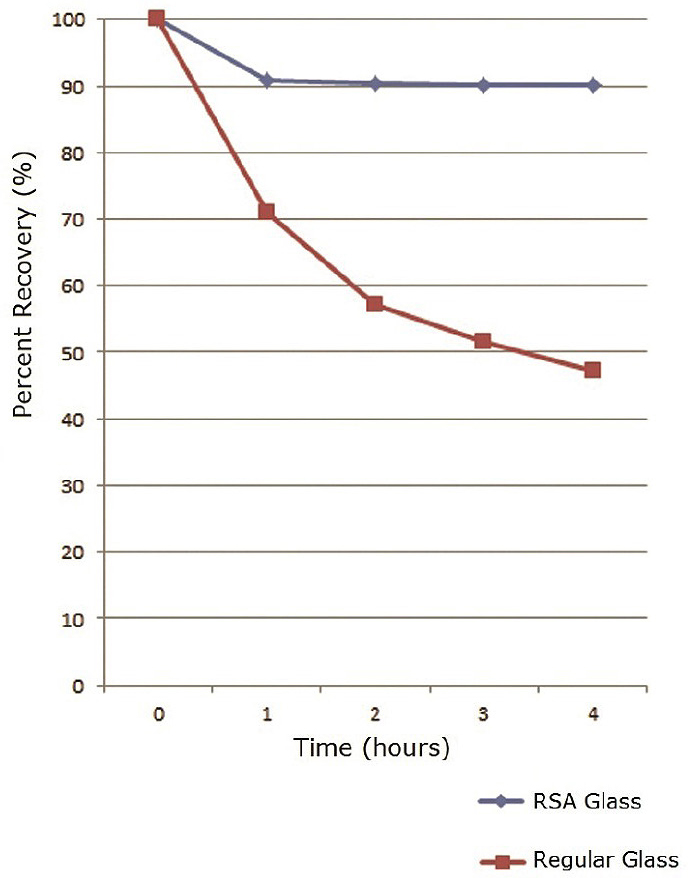
The peak area of cationic test solute, cetylpyridinium chloride was found to drastically decrease over time using conventional Type 1 Class A, borosilicate glass autosampler vials. In contrast, the RSA™ glass vial percent recovery leveled off quickly and did not drop below 90%. This is because RSA glass vials have been manufactured with virtually no surface hydroxyl groups which cause the adsorption and quantitative loss.
The chart below shows that sample loss increases within the first 4 hours with standard vials but levels off quickly with RSA vials for more reliable results.
 Method Conditions
Method Conditions
Tested Item: RSA™, Glass Vials and AQR Screw Caps
Specifications: Reduced Surface Activity Glass, Clear, 2ml, Write On, Screw Top Vials and Screw Caps
Catalog No.: 9509S-1WCP-RS
Column: Diamond Hydride™, 4μm, 100Å
Catalog No.: 70000-7.5P
Mobile Phase: 50:50 DI Water / Acetonitrile with 0.1% Formic Acid
Flow rate: 1.0mL / minute
Sample Preparation: 5.0ppm cetylpyridinium chloride reference standard in DI water diluent. Portions of the same sample were transferred to the two vial types and injected into an HPLC at time intervals of 1 hour for 4 total hours. Peak areas were recorded and compared to initial injections to calculate percent recovery.
Notes: Time dependence studies are important for several reasons. If multiple injections were made, the analyst would observe different peak areas at different times of injection, leading to poor precision. Identifying the cause may be difficult, as one may attribute it to analyte degradation or another issue. This takes up valuable lab time, resources and costs for investigation and troubleshooting.
Attachment
Basic compound adsorption study with time dependence in vials pdf Download File


