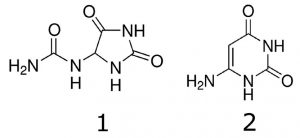
Allantoin and 6-Aminouracil
The presented method for analysis of UA metabolites in human Urine is very simple and does not require Mobile Phase additives or pre-Column derivatization. The peaks are symmetrical and the MS signal is not diminished as is the case when Ammonium Formate or Acetate is used as the Mobile Phase additive.
The analyzed metabolites are signature end products for UA degradation in the presence of oxidants and can be used as biomarkers for different disease states.
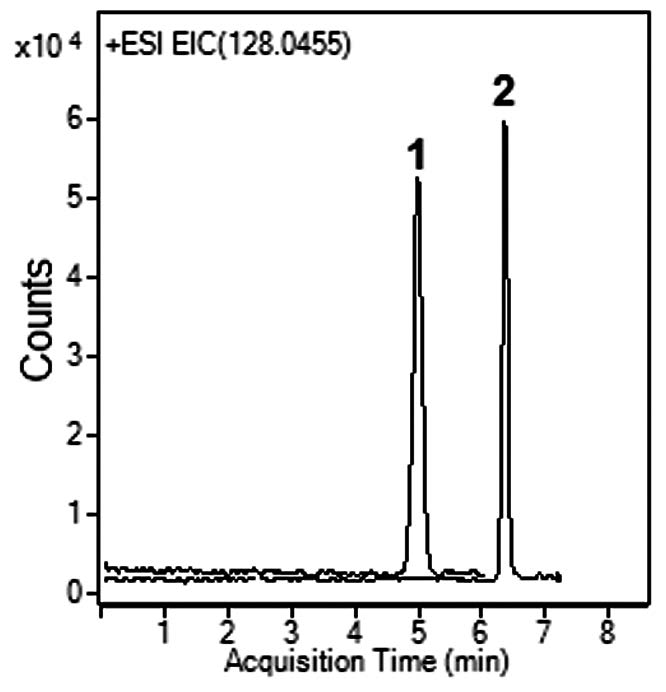
Peaks:
1. Allantoin 159.0513 m/z [M+H]+
2. 6-Aminouracil 128.0455 m/z [M+H]+
Method Conditions
Column: Cogent Diol™, 4μm, 100Å
Catalog No.: 40060-15P-3
Dimensions: 3.0 x 150mm
Mobile Phase:
—A: DI Water / 0.1% Formic Acid (v/v)
—B: Acetonitrile / 0.1% Formic Acid (v/v)
Gradient:
| Time (minutes) | %B |
| 0 | 95 |
| 1 | 95 |
| 6-7 | 30 |
| 8 | 95 |
Flow rate: 0.4 mL/minute
Detection: ESI – POS – Agilent 6210 MSD TOF Mass Spectrometer
Injection vol.: 1μL
Sample Preparation: 200 µL of Urine sample was centrifuged at 14,000 rpm for 10 minutes two times. The supernatant was diluted 1: 2 with Acetonitrile and centrifuged again at 14,000 rpm for 5 minutes. The filtrate was injected into LC-MS.
t0: 0.9 minutes
Note: Humans lack the enzyme (Uricase) which converts UA to Allantoin. However the compound still can be detected in human Urine samples. UA can react with an oxidant in a step reaction and the end product is Allantoin. When UA is degraded, it can’t be renewed. The presence of Allantoin in Urine is an indicator of a disease state (oxidative stress, renal failure, diabetes, etc.). The Allantoin/UA ratio is also elevated in Down syndrome and chronic lung disease. 6-Aminouracil is a direct product of reaction of UA with nitric oxide, which has a role in cardiovascular disease.
Attachment
No 290 Uric Acid Metabolites pdf 0.2 Mb Download File


