Simultaneous Separation using a Capillary Column
The studied analytes comprised some very Hydrophobic and Hydrophilic Peptides with diverse isoelectric points (pI) and thus served as useful probes to explore the potential of this App Note in this field of Bioanalysis.
The figure shows how these Peptides of widely disparate physicochemical properties could be separated in a single run. With these capabilities, laboratories can be more efficient and increase throughout by not having to perform separate analyses for both Hydrophobic and Hydrophilic Peptides using different Columns.
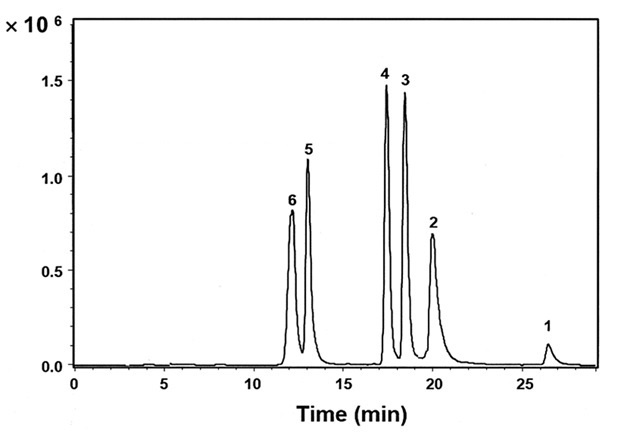
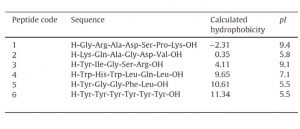
Peaks:
1. H-Gly-Arg-Ala-Asp-Ser-Pro-Lys-OH
2. H-Lys-Gln-Ala-Gly-Asp-Val-OH
3. H-Tyr-Ile-Gly-Ser-Arg-OH
4. H-Trp-His-Trp-Leu-Gln-Leu-OH
5. H-Tyr-Gly-Gly-Phe-Leu-OH
6. H-Tyr-Tyr-Tyr-Tyr-Tyr-Tyr-OH
Method Conditions:
Column: Cogent Diamond Hydride™ , 4um, 100A R&D Capillary Column
Dimensions: 0.3mm x 150mm
Solvents:
—A: DI Water / 0.5% Formic Acid (v/v)
—B: Acetonitrile / 0.5% Formic Acid (v/v)
Gradient:
| Time (minutes) | %B |
| 0 | 90 |
| 5 | 90 |
| 10 | 70 |
| 20 | 60 |
| 20.1 | 30 |
| 30 | 30 |
Injection Volume: 0.1µL
Flow Rate: 4µL / minute
Detection: ESI – POS – Agilent 6210 MSD TOF Mass Spectrometer
Sample Preparation: 10µg / mL of each Peptide in 50% Solvent A / 50% Solvent B.
Notes: The data and discussion presented here are delineated in greater detail in a full research article. Reference: R.I. Boysen, Y. Yang, J. Chowdhury, M.T. Matyska, J.J. Pesek, M.T.W. Hearn, “Simultaneous separation of hydrophobic and hydrophilic peptides with a silica hydride stationary phase using aqueous normal phase conditions.” J. Chromatogr. A, 2011, 1218, 8021–8026.
Attachment
No 371 Hydrophobic and Hydrophilic Peptides Analyzed with HPLC pdf 0.3 Mb Download File


