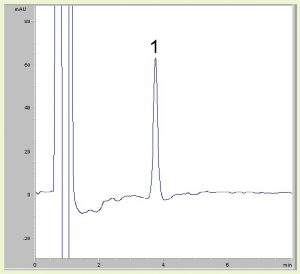
Excellent separation from matrix excipients
Pharmaceutical formulations may contain a number of excipients that can potentially interfere with a chromatographic analysis. However, using the Cogent Bidentate C8 Column, the DHEA peak is well-resolved from the others, the latter of which all elute as a large peak in the solvent front portion of the run. The API is hydrophobic and therefore retains well in the Reversed Phase mode. The use of this Column and associated conditions may be suitable for the routine assay analysis of DHEA capsule formulations.
Peak:
Dehydroepiandrosterone (DHEA)
Method Conditions
Column: Cogent Bidentate C8™, 4µm, 100Å
Catalog No.: 40008-75P
Dimensions: 4.6 x 75 mm
Solvents:
– A: 90% Water/ 10% Acetonitrile/ 0.1% Formic Acid (v/v)
– B: Acetonitrile/ 0.1% Formic Acid (v/v)
Mobile Phase: 60% A/ 40% B
Injection vol.: 5µL
Flow rate: 1 mL/minute
Detection: UV 205 nm
Sample: 500 mg strength DHEA capsule contents were added to a 50 mL volumetric flask containing a portion of Methanol diluent. The contents were sonicated for 10 min, diluted to mark, and mixed thoroughly. Then a portion was filtered (0.45µm, nylon) for injections.
Notes: DHEA is a metabolic precursor to various sex hormones. It can be used in hormone replacement therapy. Evidence for some other purported benefits such as promotion of muscle growth, memory improvement, and anti-senescence effects is currently lacking.
Attachment
No 364 Dehydroepiandrosterone DHEA Capsule.pdf 0.1 Mb Download File


