Analysis of the API and Degradants
A Sensitive, Selective, and rapid LCMS Method was developed for the simultaneous Quantification of Clopidogrel (Plavix®) and degradants.
This Method can be used for the determination of Clopidogrel in commercial tablets for Quality Control with an application for content uniformity test. The Method is also stability-indicating as it is suitable for the determination of the API in the presence of its degradation products under all stress conditions using HCL, NaOH, light and Hydrogen Peroxide.
Peaks:
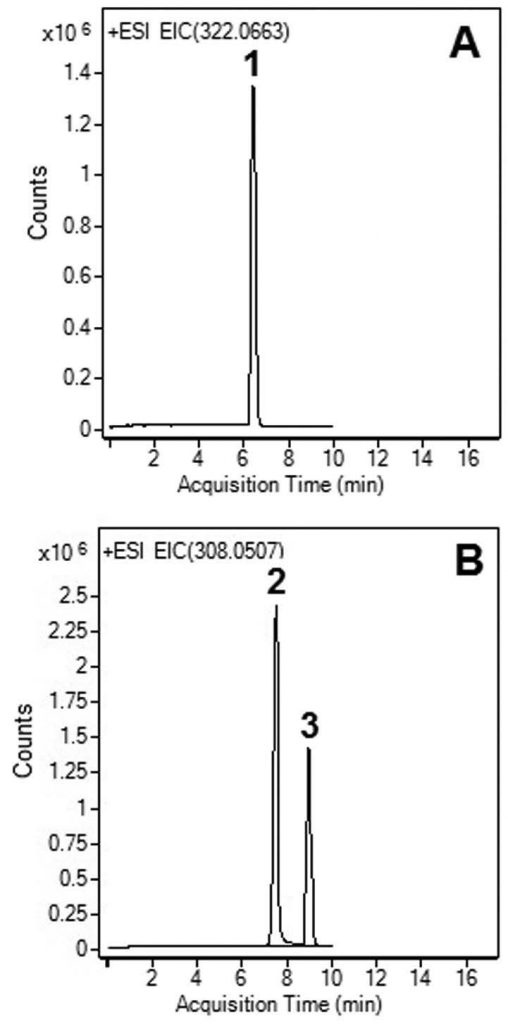
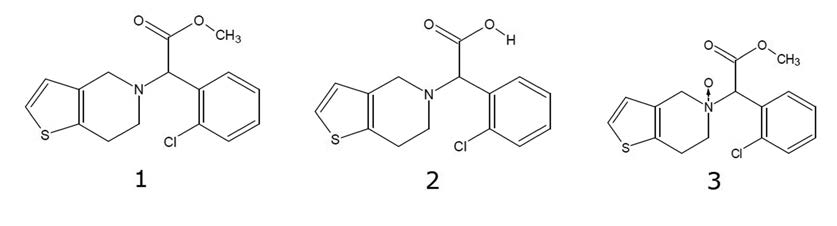
1. API: Clopidogrel, m/z 322.0663 [M+H]+
2. Degradant: Clopidogrel Acid, m/z 308.0507 [M+H]+
3. Degradant: Clopidogrel N-oxide, m/z 338.02 [M+H]+
Method Conditions
Column: Cogent Diamond Hydride™, 4μm, 100Å
Catalog No.: 70000-15P-2
Dimensions: 2.1 x 150mm
Mobile Phase:
—A: DI Water / 0.1% Formic Acid (v/v)
—B: Acetonitrile / 0.1% Formic Acid (v/v)
Gradient:
| Time (minutes) | %B |
| 0 | 95 |
| 2 | 95 |
| 7 | 60 |
| 8 | 95 |
Temperature: 25˚C
Post Time: 3 minutes
Injection vol.: 1μL
Flow rate: 0.4mL / minute
Detection: ESI – POS – Agilent 6210 MSD TOF Mass Spectrometer
Sample Preparation: 50mg strength Plavix® tablet was ground and diluted in 50:50 Solvent A / Solvent B mixture to 50mL. The solution was sonicated and filtered through a 0.45μm Nylon Syringe Filter (MicroSolv Tech Corp.).
t0: 0.9 minutes
Figures:
—Fig. A: Non-degraded extract: The stock solution was diluted 1:10 with 50:50 Solvent A / Solvent B mixture. Only the API peak is observed.
—Fig. B: Base degradation with heating: The stock solution was diluted 1:10 with 50:50 1N NaOH / Acetonitrile mixture and then heated at 85°C for 30 minutes. The API peak is no longer observed but degradants (Peak 2 and 3) are now present.
Attachment
No 258 Forced Degradation of Clopidogrel Analysis LC-MS pdf 0.3 Mb Download File