Quick Analysis without Ion Pair Agents
The USP Assay Method for Atenolol uses Heptane Sulfonate and Dibutyl Amine in the Mobile Phase, which are slow to load and remove from a Reversed Phase HPLC Column. These reagents are added to improve the Peak Tailing, which must be not more than 2.0.
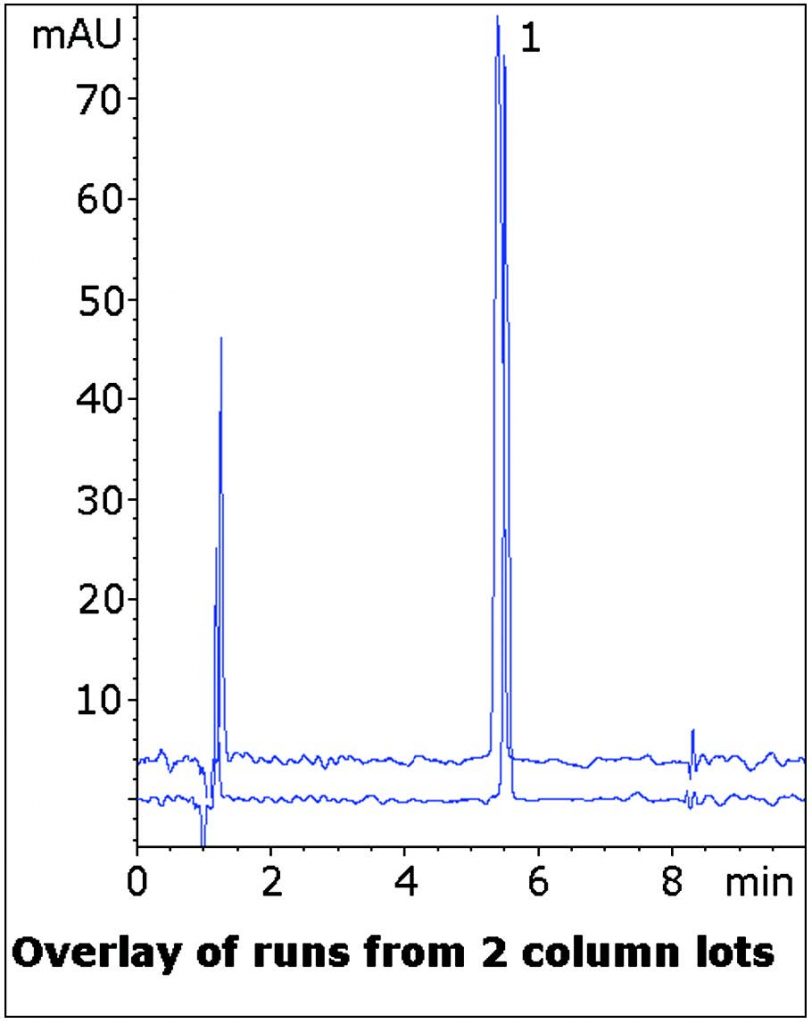
The Method in this Application Note is an improvement over the official USP Method as the Peak produced is very Symmetrical without the use of Ion Pair Agents. Also, the Method is LCMS compatible if transfer is desired. Data from two Column lots is shown to illustrate lot-to-lot consistency of this Method.
Peak:
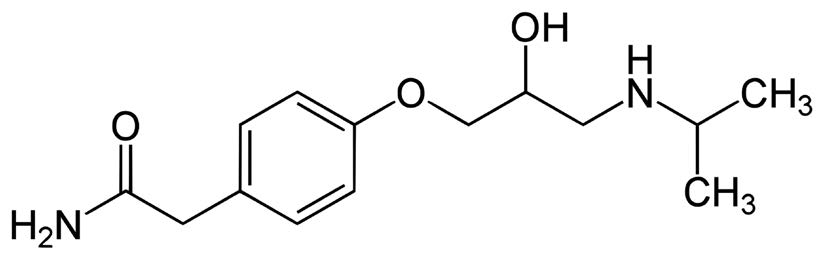
Atenolol
Method Conditions
Column: Cogent Diamond Hydride™, 4μm, 100Å
Catalog No.: 70000-7.5P
Dimensions: 4.6 x 75mm
Mobile Phase:
—A: DI Water with 0.1% Formic Acid (v/v)
—B: Acetonitrile with 0.1% Formic Acid (v/v)
Gradient:
| Time (minutes) | %B |
| 0 | 90 |
| 1 | 90 |
| 6 | 40 |
| 7 | 90 |
Post Time: 3 minutes
Injection vol.: 2μL
Flow rate: 1.0mL / minute
Detection: UV @ 225nm
Sample Preparation: 25mg strength Atenolol tablet was ground and added to a 25mL volumetric flask. A portion of 50:50 Solvent A / Solvent B diluent was added and the flask was sonicated 10 minutes. It was then diluted to mark and mixed. A portion was filtered with a 0.45μm Nylon Syringe Filter (MicroSolv Tech Corp.) and diluted 1:10.
t0: 0.9 minutes
Note: Atenolol is a selective ß1 receptor antagonist. It is used to treat various cardiovascular diseases such as hypertension. It is available by prescription under the trade name Tenormin®.
Attachment
No 250 Atenolol Tablet Analyzed with HPLC pdf 0.4 Mb Download File