Reducing Tailing for Venlafaxine with HPLC
The USP assay Method for Venlafaxine capsules uses Triethylamine and Phosphoric Acid in the Mobile Phase, both of which are incompatible with LC-MS. The system suitability for Venlafaxine tailing factor is 2.0, indicating the compound has a tendency for tailing. Here a sharp symmetrical peak is observed using Formic Acid. Data from two Column lots is shown to illustrate reproducibility.
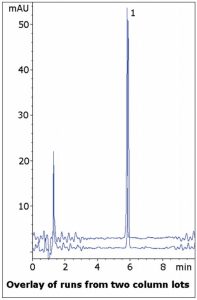
 Peak:
Peak:
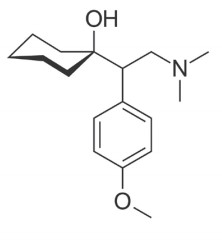
Venlafaxine
Method Conditions
Column: Cogent Diamond Hydride™, 4µm, 100Å
Catalog No.: 70000-7.5P
Dimensions: 4.6 x 75mm
Mobile Phase:
—A: DI Water / 0.1% Formic Acid (v/v)
—B: Acetonitrile / 0.1% Formic Acid (v/v)
Gradient:
| Time (minutes) | %B |
| 0 | 95 |
| 1 | 95 |
| 6 | 50 |
| 7 | 95 |
Post Time: 3 minutes
Injection vol.: 1µL
Flow rate: 1.0mL / minute
Detection: UV @ 226nm
Sample Preparation: 75mg strength Effexor® Extended Release capsule contents were added to a 25mL volumetric flask. A portion of 50/50 Solvent A / Solvent B diluent was added and the flask was sonicated 10 minutes. It was then diluted to mark and mixed. A portion was filtered with a 0.45µm Nylon Syringe Filter (MicroSolv Tech Corp.) and diluted 1:50.
t0: 0.9 minutes
Note: Venlafaxine is a serotonin-norepinephrine reuptake inhibitor used to treat various depressive and anxiety disorders. It is currently marketed by Pfizer as Effexor®. It is a phenethylamine and shares structural similarities with other compounds in this class, such as amphetamine, methamphetamine, and MDMA.
Attachment
No 237 Effexor Venlafaxine Capsule Analyzed with HPLC pdf 0.4 Mb Download File


