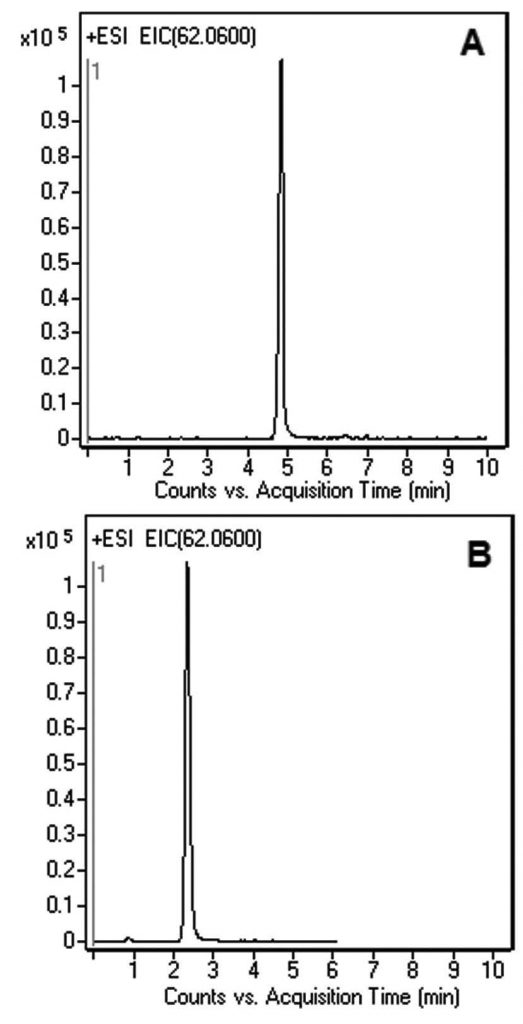
Retention & Analysis of Hydrophilic, Ethanolamine from Plasma
In this Application Note, the Chromatograms show Ethanolamine sufficiently retained using either the Gradient or Isocratic Methods. The advantages of using these Methods are fast equilibration between runs when Gradient analysis is required and due to the high organic content of the Mobile Phase, the Ionization efficiency of MS Detector produces increased sensitivity when compared to Methods with a high Water content Mobile Phase.
Peak:
Ethanolamine [M + H]+ 62.0600 m/z
Method Conditions
Column: Cogent Diamond Hydride™, 4μm, 100Å
Catalog No.: 70000-15P-2
Dimensions: 2.1 x 150mm
Mobile Phase:
—Gradient Figure A
—A: 50:50 DI Water / Isopropanol with 0.1% Formic Acid (v/v)
—B: 97:3 Acetonitrile / DI Water with 0.1% Formic Acid (v/v)
—Isocratic Figure B
—50:50 Solvent A / Solvent B
Gradient:
| Time (minutes) | %B |
| 0 | 80 |
| 4 | 40 |
| 6 | 40 |
| 7 | 80 |
Post Time: 3 minutes
Injection vol.: 1μL
Flow rate: 0.4mL / minute
Detection: ESI – POS – Agilent 6210 MSD TOF Mass Spectrometer
t0: 0.9 minutes
Note: Ethanolamine is bi-functional, containing both a primary amine and a primary alcohol. Colorless, viscous and with an odor reminiscent of ammonia, its derivatives are widespread in nature, including: modulating several animal and plant physiological processes such as seed germination, plant–pathogen interactions, chloroplast development and flowering. It is also a precursor and when combined with arachidonic acid the endocannabinoid Anandamide is formed.
Attachment
No 210 Ethanolamine Analyzed with LCMS pdf 0.3 Mb Download File