Formulation of Guaifenesin and Dextromethorphan HBr
Click HERE for Column Ordering Information.
This Method shows the separation of two active ingredients in a formulation of Guaifenesin and Dextromethorphan. Dextromethorphan often exhibits tailing in many HPLC methods, but here an excellent Peak shape is observed.
This Method is suitable for routine assay of these compounds in combination formulations. A change in wavelength was used in order to obtain similar Peak heights between the two compounds, since their concentrations differ significantly.
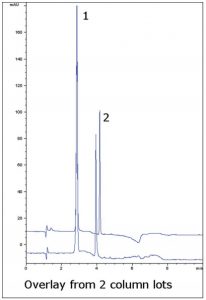
 Peaks:
Peaks:
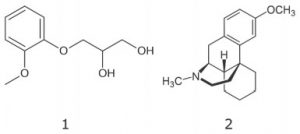
1. Guaifenesin
2. Dextromethorphan
Method Conditions
Column: Cogent Phenyl Hydride™, 4µm, 100Å
Catalog No.: 69020-75P
Dimensions: 4.6 x 75mm
Mobile Phase:
—A: DI Water / 0.1% TFA (v/v)
—B: Acetonitrile / 0.1% TFA (v/v)
Gradient:
| Time (minutes) | %B |
| 0 | 20 |
| 1 | 20 |
| 5 | 90 |
| 6 | 20 |
Temperature: 35˚C
Post Time: 4 minutes
Flow rate: 1.0mL / minute
Detection: UV @ 254nm (0-3.5 minutes), 215nm (3.5-10 minutes)
Injection vol.: 2µL
Sample Preparation: Coricidin High Blood Pressure (HBP) Day® tablet containing 200mg Guaifenesin and 10mg Dextromethorphan HBr was ground and added to a 50mL volumetric flask. A portion of 50:50 Solvent A / Solvent B was added and the flask was sonicated 10 minutes. Then it was diluted to mark and mixed well. A portion was filtered with a 0.45µm Nylon Syringe Filter (MicroSolv Tech Corp.). This filtrate was used for injections. The Peak identities were confirmed with individual standards.
t0: 0.9 minutes
Note: Guaifenesin is an expectorant agent and Dextromethorphan is an antitussive. This particular cold and cough formulation is designed for individuals with high blood pressure.
Attachment
No 206 Coricidin HBP Day Tablet Analyzed with HPLC pdf 0.3 Mb Download File


