Rapid, Reliable Isocratic Method for this API
The USP Assay Monograph Method for Meloxicam uses a phosphate based Mobile Phase. In this application note, a 0.1% Formic Acid additive is chosen in lieu of phosphates in order to be LCMS compatible.
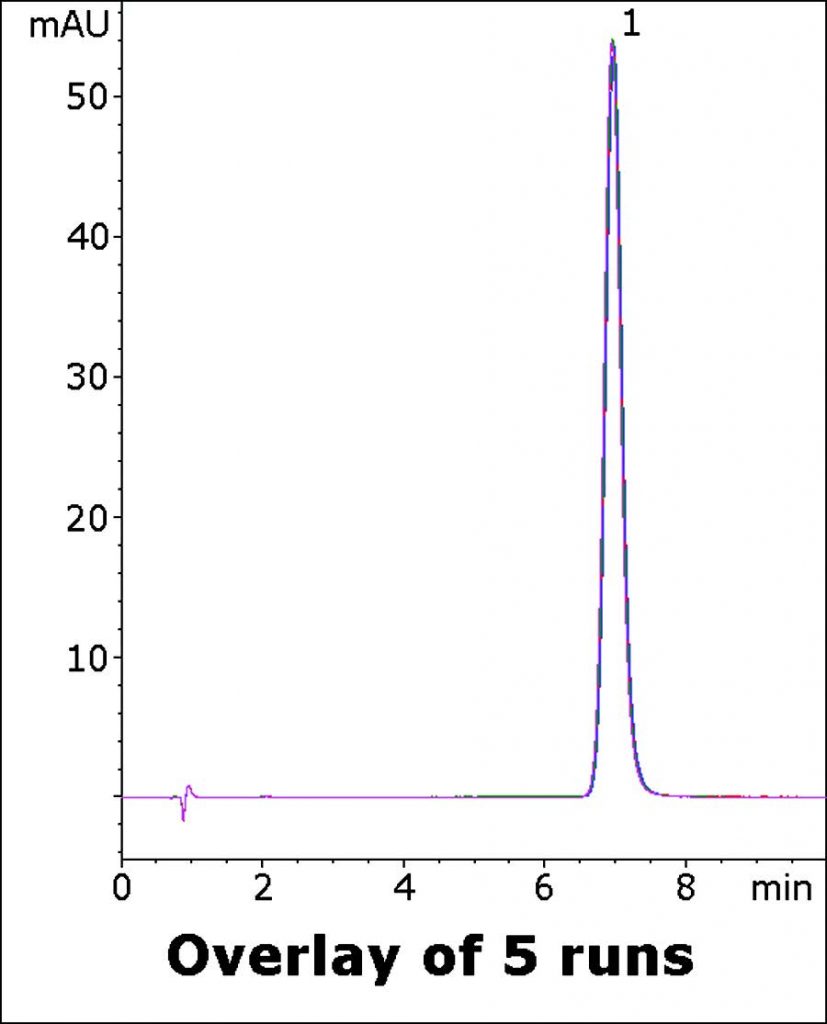
The System Suitability requirement is not more than 2.0 for Tailing factor. Highly Symmetrical Peaks are obtained with this Method which is easy to perform and is suitable for routine Assay of Meloxicam Tablets. Five Chromatograms are overlaid below to demonstrate the Robustness and Precision of this Method.
Peak:
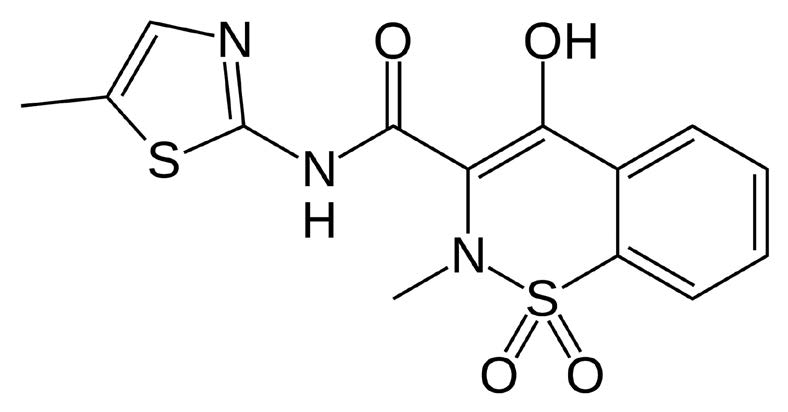
Meloxicam
Method Conditions
Column: Cogent UDC-Cholesterol™, 4μm, 100Å
Catalog No.: 69069-7.5P
Dimensions: 4.6 x 75mm
Mobile Phase: 60:40 DI Water / Acetonitrile with 0.1% Formic Acid (v/v)
Injection vol.: 10μL
Flow rate: 1.0mL / minute
Detection: UV @ 362nm
Sample Preparation:
—Stock Solution: 15mg strength Meloxicam tablet was ground and added to 50mL volumetric flask containing a portion of the Mobile Phase as diluent. It was sonicated 10 minutes and diluted to mark. A portion was filtered through a 0.45μm Nylon Syringe Filter (MicroSolv Tech Corp.).
—Working Solution: The stock solution was diluted 1:10 with the Mobile Phase diluent.
Note: Meloxicam is a non-steroidal anti inflammatory drug which is used for treatment of various inflammatory illnesses, including osteoarthritis and rheumatoid arthritis. It acts by inhibition of Cyclooxygenase, which catalyzes the conversion of Arachidonic Acid to Prostaglandin H2. In this manner, it interferes with the synthesis of prostaglandins, which mediate inflammation.
Attachment
No 192 Meloxicam Tablet Analyzed with HPLC pdf 0.3 Mb Download File