API well separated from degradation product
Using this Simple Linear Gradient, Atorvastatin is well separated from its main degradation Product. The degradant is formed under acidic conditions and therefore its Separation is important for Stability Indicating Methods for pharmaceutical formulations of Atorvastatin. For Methods requiring non-degrading conditions, it is important to choose diluent and Mobile Phase conditions with pH sensitivity in mind.
As such, Ammonium Acetate was chosen for the diluent and Mobile Phase additives in order to avoid any degradation from these sources. Data obtained under non-degrading conditions is shown in Figure A. An overlay of five consecutive runs of the degraded extract is shown in Figure B. The Repeatability of the Method is demonstrated by the consistent retention time (%RSD of 0.10).
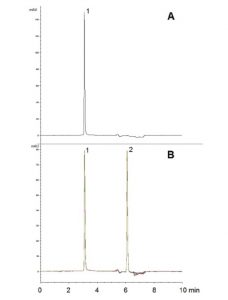
 Peaks:
Peaks:
1. Atorvastatin
2. Degradant
Method Conditions
Column: Cogent Bidentate C18™, 4μm, 100Å
Catalog No.: 40018-15P
Dimensions: 4.6 x 150mm
Mobile Phase:
—A: DI Water / 10mM Ammonium Acetate
—B: 90% Acetonitrile / 10% DI Water / 10mM Ammonium Acetate
Gradient:
| Time (minutes) | %B |
| 0 | 40 |
| 5 | 100 |
| 6 | 40 |
Injection vol.: 2μL
Flow rate: 0.8mL / minute
Detection: UV @ 248nm
Sample Preparation:
—A: Atorvastatin tablet extract
—B: Degraded Atorvastatin tablet extract
40mg strength tablet ground was placed in 100mL volumetric flask. 50mL of Mobile phase B was added to flask. Sample was vortexed for 5 minutes, sonicated for 5 minutes, then diluted to mark with Solution A. A portion was filtered through a 0.45μm Nylon Syringe Filter (MicroSolv Tech Corp.). Final Working Solution was then diluted 4x in Sample A) 50:50 A / B or Sample B) 1.0 N HCL in 50:50 A / B.
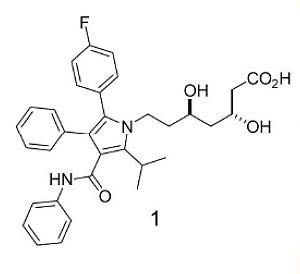
Note: Atorvastatin is a competitive inhibitor of 3-Hydroxy-3-Methlyglutaryl Co-enzyme A (HMGCoA) reductase, which catalyzes the rate-limiting step in Cholesterol biosynthesis. As such, Atorvastatin is used to reduce plasma levels of low-density lipoprotein (LDL) Cholesterol, which are known to contribute to the development of Atherosclerosis.
Attachment
Atorvastatin Drug Product Analyzed with HPLC pdf Download File


