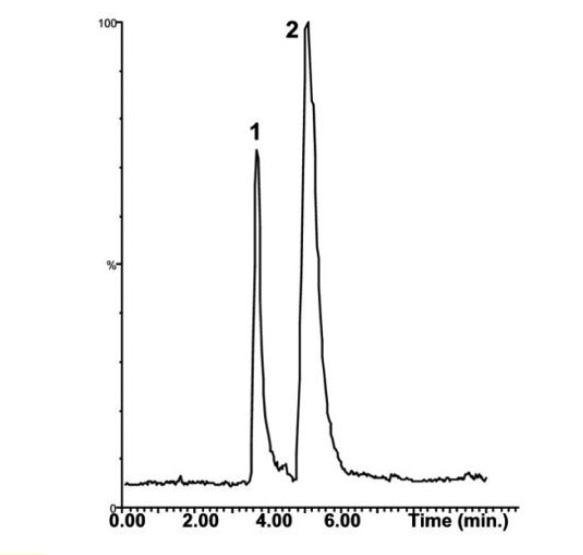
Retention of highly polar, water soluble antibiotic
Tobramycin is a very polar compound that lacks a sufficient chromophore, and therefore is difficult to analyze using Reversed Phase HPLC with UV detection. In this Application Note, the Method below retains Tobramycin and Separates it Uracil which is used as a Solvent Front Marker.
Using this Method, Reversed Phase Method is possible and successful due to the Ion Interaction between the Stationary Phase’s terminal Carboxylic Acid (wcx) group and Tobramycin.
Peaks:
1. Uracil (m/z 113)
2. Tobramycin (m/z 468)
Method Conditions
Column: Cogent UDA™, 4μm, 100Å
Catalog No.: 40031-75P
Dimensions: 4.6 x 75mm
Mobile Phase: 80:20 DI Water / Acetonitrile with 0.5% Formic Acid
Injection vol.: 1μL (internal loop)
Flow rate: 0.5mL / minute
Detection: APCI+ Single Ion Monitoring Mass Spectrometer.
Sample Preparation: 1mg / L of each dissolved in DI Water.
Note 1: Aminoglycoside drugs are difficult to retain on conventional HPLC columns due to their highly polar characteristics and Ion Pair Reagents are typically used to induce retention but they are not preferred because they interfere with MS detection. Above Method does not use any Ion Pair Agents.
Note 2: Tobramycin is a water-soluble aminoglycoside antibiotic purified from the fermentation of the Actinomycete Streptomyces Tenebrarius. It is used in a variety of pharmaceutical applications: ophthalmic suspensions and ointments, such as TobraDex® (Alcon Inc., Fort Worth, TX), inhalation solutions such as TOBI® (Chiron Corporation, Woodstock, IL), and intravenous administrations such as Tobramycin Sulfate Injection (Eli Lilly and Company, Indianapolis, IN).
Attachment
Tobramycin Analyzed with LCMS pdf Download File