Separation of API from Acid Degradation Products
This Method illustrates the capabilities to separate several Glipizide Degradants that are formed under acidic conditions. Good separation is obtained as well as Sharp, Symmetric Peak shapes for each compound.
The Retention times show good repeatability as well, as demonstrated in the Figure overlays. Also, the Mobile Phase Solvents are LCMS compatible, which expands the potential applications of the Method. Finally, Gradient Equilibration is fast and requires only 1 Column volume.
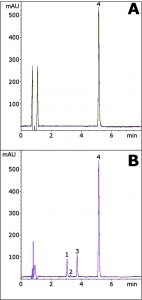
Peaks:
1. Degradant
2. Degradant
3. Degradant
4. Glipizide
Method Conditions
Column: Cogent Bidentate C18™, 4µm, 100Å
Catalog No.: 40018-75P
Dimensions: 4.6 x 75mm
Mobile Phase:
—A: DI Water / 0.1% Formic Acid
—B: Acetonitrile / 0.1% Formic Acid
Injection vol.: 10µL
Flow rate: 1mL / minute
Detection: UV @ 225nm
Sample Preparation:
—Figure. A (Non-Degraded): 0.1mg / mL Glipizide in Methanol diluent.
—Figure. B (Acid Degradation): 0.1mg / mL Glipizide in 50:50 Methanol /1N HCl diluent. Sample was heated at 85°C for 1 hour.
t0: 0.9 minutes
Note: Glipizide is an anti-diabetic sulfonylurea drug, sold under the trade name Glucotrol®.
Attachment
No 165 Forced Degradation of Glipizide with HPLC pdf 0.3 Mb Download File


