USP Assay Method an Antibiotic Prodrug
The USP Assay Method for Cefpodoxime Proxetil specifies a Resolution of not less than 2.5 between the two epimers of the Prodrug. Using this Method, the average Resolution was calculated to be 2.8. In addition, the R-epimer tailing factor must be not more than 1.5 where this Method meets this requirement with a tailing factor of 1.2. Lastly, this Method also shows good Repeatability with a Retention Time %RSD from five runs of 0.2%.
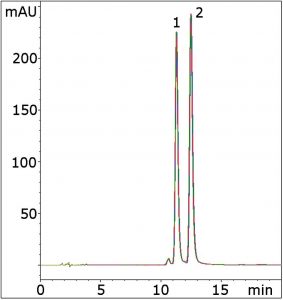
Peaks:
1. Cefpodoxime Proxetil, S-epimer
2. Cefpodoxime Proxetil, R-epimer
Method Conditions
Column: Cogent Bidentate C18™, 4μm, 100Å
Catalog No.: 40018-25P
Dimensions: 4.6 x 250mm
Mobile Phase: 60% 20mM Ammonium Acetate / 40% Acetonitrile
Temperature: 30˚C
Injection vol.: 20μL
Flow rate: 1.0mL / minute
Detection: UV @ 235nm
Sample Preparation:
—Stock Solution: A 200 mg strength Cefpodoxime Proxetil tablet was ground and added to a 100mL volumetric flask. The flask was diluted to mark with the Mobile Phase and sonicated. A portion was then filtered with a 0.45µm Nylon Syringe Filter (MicroSolv Tech Corp.).
—Working Solution: 100μL of the Stock Solution was diluted with 900μL of the Mobile Phase.
t0: 1.9 minutes
Note: Cefpodoxime Proxetil is the Prodrug of Cefpodoxime, which is formed by hydrolysis of the Prodrug ester bond. Cefpodoxime is a Cephalosporin Antibiotic used to treat conditions such as Acute Otitis Media, Pharyngitis, and Sinusitis. It is marketed by Pfizer as Vantin®, but generic versions are also available.
Attachment
No 149 Cefpodoxime Proxetil Analyzed with HPLC pdf 0.3 Mb Download File


