Impurities Method with Improved Peak Shape
Click HERE for Column Ordering Information.
Celecoxib is Separated from an Impurity in this simple Gradient Method. The Peak shape obtained for this API shows good Peak Symmetry. In addition, the Method shows good repeatability from run to run. The API Retention time %RSD from five runs was 0.025%.
Figure A shows the excellent Precision with five Chromatograms overlayed. Figure B shows a zoom-in view so that the impurity peak can be seen clearly. Another benefit of the Method is that the Mobile Phase is MS compatible.
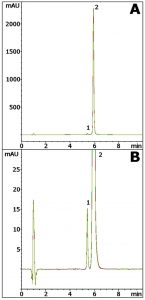
Peaks:
1. Impurity
2. Celecoxib
Method Conditions
Column: Cogent Phenyl Hydride™, 4μm, 100Å
Catalog No.: 69020-7.5P
Dimensions: 4.6 x 75mm
Mobile Phase:
—A: DI Water / 0.1% Formic Acid
—B: 97% Acetonitrile / 3% DI Water / 0.1% Formic Acid
Gradient:
| Time (minutes) | %B |
| 0 | 30 |
| 5 | 60 |
| 6 | 30 |
Temperature: 35˚C
Post Time: 4 minutes
Flow rate: 1.0mL / minute
Detection: UV @ 254nm
Injection vol.: 1μL
Sample Preparation: A 200mg strength Celebrex® (Celecoxib) capsule was ground and added to a 100mL volumetric flask. 50mL Methanol was added and the solution was sonicated. The flask was diluted to mark with Solvent A and a portion was filtered with a 0.45µm Nylon Syringe Filter (MicroSolv Tech Corp.).
t0: 0.9 minutes
Note: Celecoxib is a selective COX-2 inhibitor used for treatment of acute pain and inflammation due to conditions such as Arthritis.
Attachment
No 148 Celecoxib Analyzed with HPLC pdf 0.3 Mb Download File


