Separation Achieved with Shape Selectivity and Hydrophobic Interaction
This Method provides an example of Reversed Phase Separation containing a number of related compounds. The properties of the solutes are listed in the Table below.
When comparing the properties of Compounds 2 and 3, it is interesting to note that 2 is more hydrophobic than 3 but it’s Retention is shorter. The Method uses Shape Selectivity as well as Hydrophobic Interaction which may explain why the less hydrophobic compound is retained longer. In general, the more polar compounds elute earlier than the more hydrophobic species. Compound 7 has no polar functionalities at all so it is the most retained under Reversed Phase conditions.
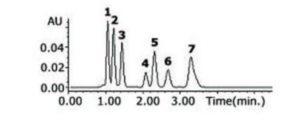
Peaks:
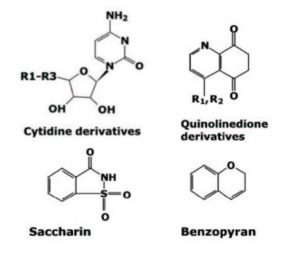
1. Cytidine-R1
2. Cytidine-R2
3. Cytidine-R3
4. Quinolinedione-R1
5. Saccharin
6. Quinolinedione-R2
7. Benzopyran
Method Conditions
Column: Cogent UDC Cholesterol™, 4μm, 100Å
Catalog No.: 69069-7.5P
Dimensions: 4.6 x 75mm
Mobile Phase: 60:40 Acetonitrile / DI Water + 0.5% Perchloric Acid
Injection vol.: 5μL
Flow rate: 1mL / minute
Detection: UV @ 270nm
Sample Preparation: 1mg / mL of each in Acetonitrile + DI Water
Table:
| Solute | Mol Wt. | B pKa | Log P |
| Cytidine-R1 | 397 | 3.73 | 1.54 |
| Cytidine-R2 | 454 | 3.73 | 3.66 |
| Cytidine-R3 | 425 | 3.73 | 2.66 |
| Quinolinedione-R1 | 536 | – | 3.46 |
| Saccharin | 536 | 5.36 | 2.96 |
| Quinolinedione-R2 | 520 | – | 2.74 |
| Benzopyran | 396 | – | 2.80 |
Attachment
Cancer Prodrug Analyzed with HPLC pdf 0.2 Mb Download File


