Robust Analysis of Amoxicillin, Antibiotic with HPLC
The USP Assay Monograph Method for Amoxicillin often yields poor Peak shapes and low Method reproducibility with L1 Columns based on ordinary Silica. In contrast, the Figures shown here illustrate the high degree of reproducibility obtained from use of the Cogent Bidentate C18 L1 Column, following the USP Assay Method.
Each figure shows a five run overlay of consecutive runs. Figure A shows the data obtained using the analyte concentration specified by the USP Assay Method. Figure B shows a lower concentration which is less likely to result in Column overload.
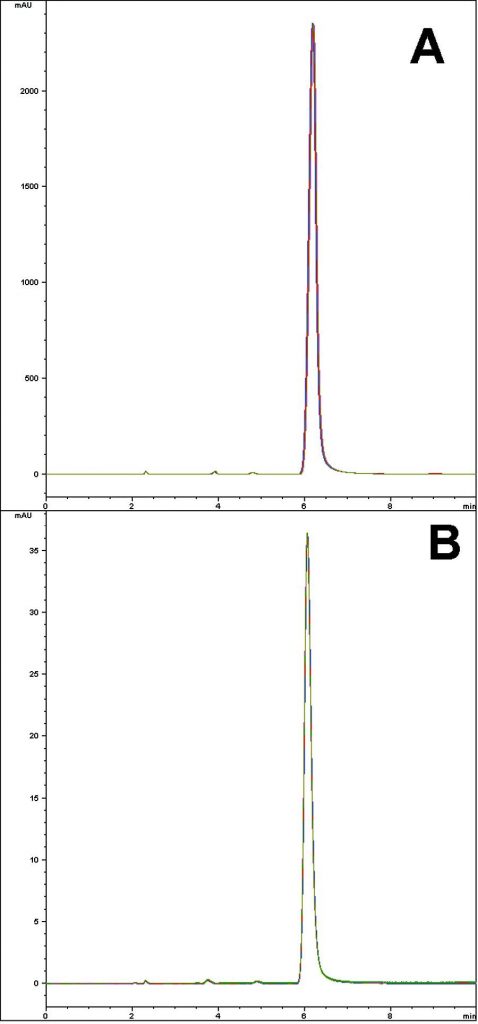
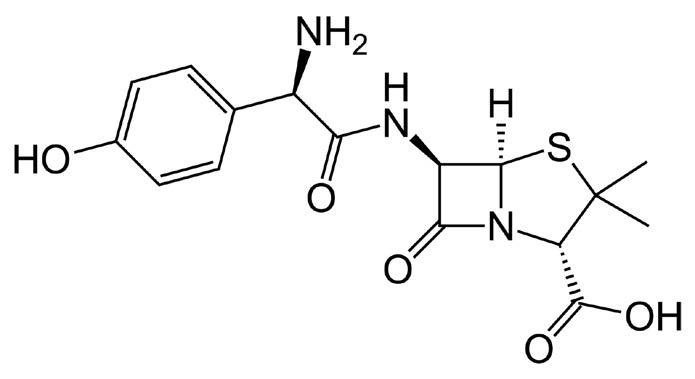
Peaks:
Amoxicillin
Method Conditions
Column: Cogent Bidentate C18™, 4μm, 100Å
Catalog No.: 40018-25P
Dimensions: 4.6 x 250mm
Mobile Phase: 3:97 Acetonitrile: Diluent (6.8 g/L monobasic Potassium Phosphate adjusted to pH 5.0 with 45% (w/w) Potassium Hydroxide)
Injection vol.: 10μL
Flow rate: 1.0mL / minute
Detection: UV @ 230nm
Sample Preparation:
—Figure A: 1.2mg / mL Amoxicillin Trihydrate USP RS in the diluent.
—Figure B: 0.012mg / mL Amoxicillin Trihydrate USP RS in the diluent.
t0: 2.3 minutes
Note: Amoxicillin is a beta-lactam antibiotic used to treat a variety of bacterial infections. Its mechanism of action is by inhibition of the synthesis of bacterial cell walls.
Attachment
No 136 Amoxicillin USP Method pdf 0.3 Mb Download File


