Antibiotic Tablet
Ciprofloxacin has several amine groups and therefore is susceptible to peak tailing in Reversed Phase methods. The USP assay method for Ciprofloxacin tablets uses Triethylamine as an Ion Pair Agent to reduce tailing and in addition, Phosphoric Acid is included in the Mobile Phase.
These conditions make the USP method incompatible with MS detection and therefore limit its applications. In this method, only Formic Acid is required in order to obtain a peak that meets the system suitability specification for the API tailing factor.
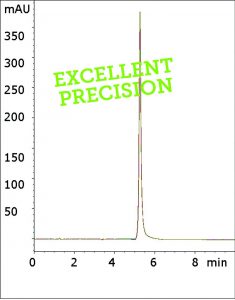
Peak:
Ciprofloxacin
Method Conditions
Column: Cogent Diamond Hydride™, 4μm, 100Å
Catalog No.: 70000-7.5P
Dimensions: 4.6 x 75mm
Mobile Phase:
—A: DI Water / 0.1% Formic Acid (v/v)
—B: Acetonitrile / 0.1% Formic Acid (v/v)
Gradient:
| Time (minutes) | %B |
| 0 | 95 |
| 1 | 95 |
| 6 | 50 |
| 7 | 95 |
Post Time: 3 minutes
Flow rate: 1.0 mL / minute
Detection: UV @ 270 nm
Injection vol.: 1μL
Sample Preparation: A 250 mg strength Ciprofloxacin tablet was ground and dissolved in 25 mL of 50% solvent A / 50% (v/v) solvent B diluent. Solution was sonicated 10 minutes and filtered through a 0.45µm Nylon Syringe Filter (MicroSolv Tech Corp.). Sample for injection was diluted 1:10 with the same diluent.
t0: 0.9 minutes
Note: Ciprofloxacin is a Fluoroquinolone antibiotic which acts by inhibition of DNA Gyrase and Topoisomerase IV. It was once used as a drug of last resort for bacterial infections, but accumulated resistance has made the drug less effective in recent years.
Attachment
No 172 Ciprofloxacin LC-MS Compatible Assay pdf 0.5 Mb Download File


