Isocratic Assay Method for a Diabetes Drug
In this study, an Isocratic, HPLC Method for Glimepiride Analysis in a tablet formulation is presented. The USP Assay Method for this drug calls for a Phosphate Buffer and is therefore not LCMS compatible. This Method uses Formic Acid instead and produces a Symmetrical, well-Retained Peak. Therefore the analysis could be applied to various LCMS studies as well if desired, such as with clinical laboratories dealing with biological extracts.
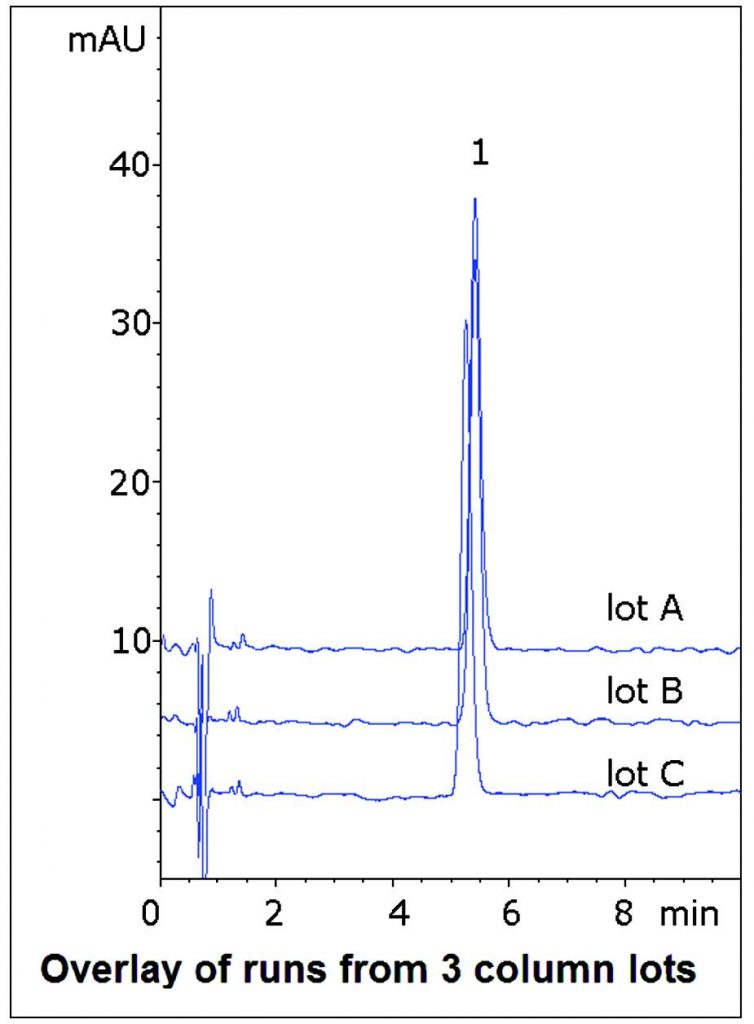
Data from three different Column lots is shown in the figure in order to demonstrate the Method Reproducibility and Robustness.
Method Conditions
Column: Cogent Bidentate C18™, 4μm, 100Å
Catalog No.: 40018-75P
Dimensions: 4.6 x 75mm
Mobile Phase: 50:50 DI Water / Acetonitrile with 0.1% Formic Acid (v/v)
Injection vol.: 5μL
Flow rate: 1.0mL / minute
Detection: UV @ 230nm
Sample Preparation: 1mg strength Glimepiride Tablet was ground and transferred to a 4mL volumetric flask. A portion of 50:50 Acetonitrile / DI Water was added and the flask was sonicated 10 minutes. It was then diluted to mark and mixed. A portion was filtered with a 0.45μm Nylon Syringe Filter (MicroSolv Tech Corp.) and diluted 1:10.
t0: 0.9 minutes
Note: Glimepiride is a sulfonylurea drug indicated to treat type 2 Diabetes Mellitus. It works by increasing insulin production in the pancreas. It is sold under trade names such as Amaryl®.
Attachment
No 253 Glimepiride Tablet Analyzed with HPLC pdf 0.4 Mb Download File