Assay for Anti-Obesity Combination Capsule & Tablet
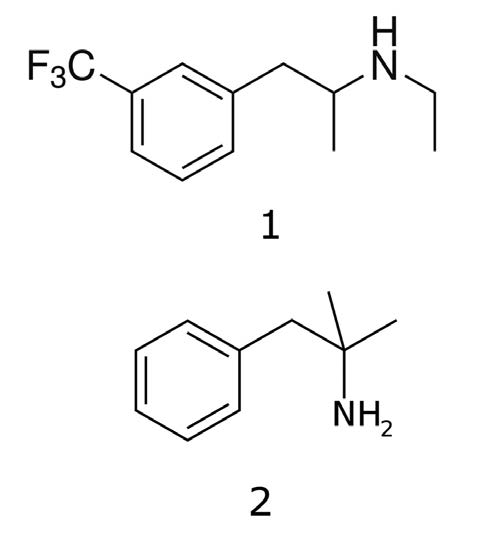
This Application Note illustrates Separation of similar compounds in the Phenethylamine class of compounds; Fenfluramine & Phentermine.
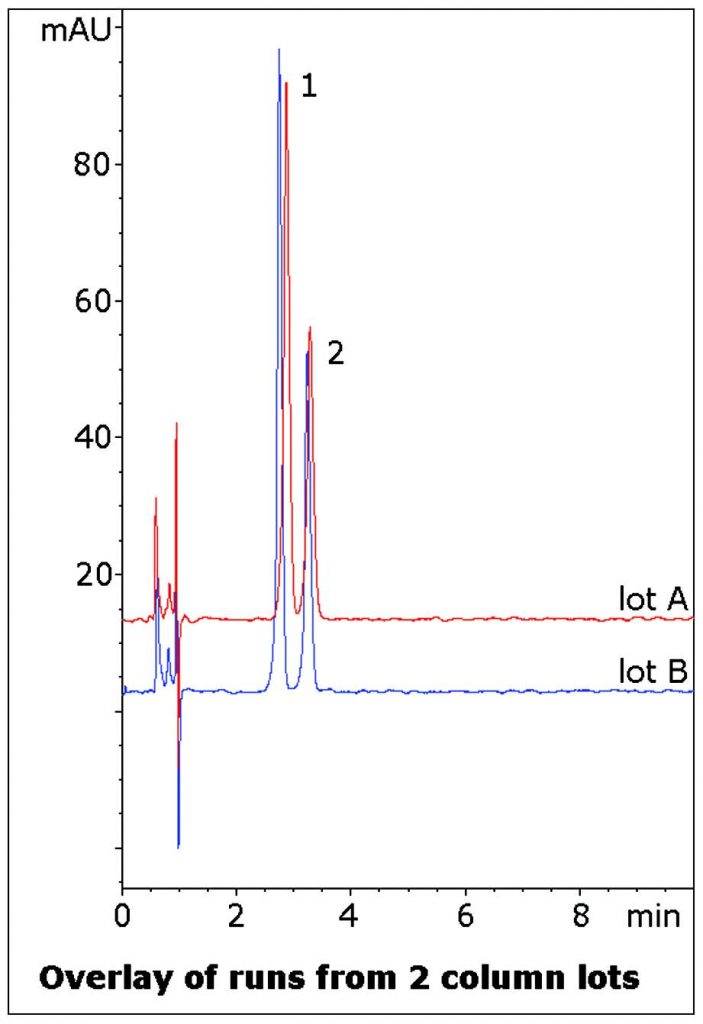
These two compounds in particular were once used as APIs in the same formulation. The data shows how these Phenethylamines can be easily Retained and Separated with an Isocratic Mobile Phase that is LCMS compatible and easy to prepare. Data from two lots is shown to illustrate the Reproducibility of the Method.
Peak:
1. Fenfluramine
2. Phentermine
Method Conditions
Column: Cogent Diamond Hydride™, 4μm, 100Å
Catalog No.: 70000-7.5P
Dimensions: 4.6 x 75mm
Mobile Phase: 25:75 DI Water / Acetonitrile with 0.1% Formic Acid (v/v)
Injection vol.: 1μL
Flow rate: 1.0mL / minute
Detection: UV @ 210nm
Sample Preparation: 30mg strength Ionamin® capsule contents were added to a 25mL volumetric flask. A portion of mobile phase diluent was added and the flask was sonicated for 10 minutes. It was then diluted to mark, mixed, and filtered with a 0.45μm Nylon Syringe Filter (MicroSolv Tech Corp.). 20mg strength Pondimin® Tablet was ground and added to another 25mL volumetric flask. A filtrate was obtained in the same manner as with Ionamin®, and the two filtrates were mixed 1:1. Individual extracts were used for peak identity confirmation.
t0: 0.9 minutes
Note: Fenfluramine and Phentermine were once sold as an anti-obesity medication, however, Fenfluramine was shown to produce serious side effects of pulmonary hypertension and heart valve problems. Phentermine does not produce these side effects.
Attachment
No 239 Fenfluramine and Phentermine Analyzed with HPLC pdf 0.4 Mb Download File