Purine Nucleotides Analyzed with HPLC- AppNote
May 17, 2012
/
/
/
/
/
Separation of Xanthosine -5’-Monophopate and Guanosine -5’-Triphosphate
This Method achieves the separation of Nucleotides XMP from GTP. The analysis of these polar compounds is achieved at high concentration of an organic Solvent as part of the Mobile Phase which provides increased Sensitivity. Retention times were very Reproducible with %RSD approximately 0.4, even when Red Blood Cell extracts were injected in between the standard samples.
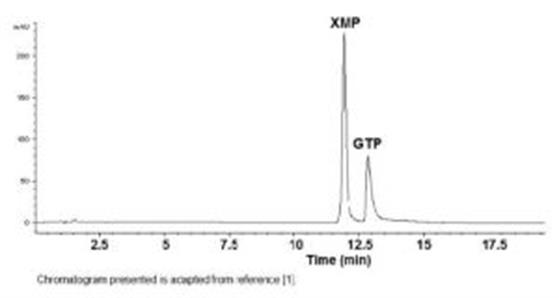
Column: Cogent Diamond Hydride™, 4µm, 100Å
Catalog No.: 70000-15P-2
Dimensions: 2.1 x 150mm
Mobile Phase:
--A: DI Water + 0.1% Ammonium Formate
--B: 90% Acetonitrile / 10% DI Water / 0.1% Ammonium Formate
Gradient:
Temperature: 25°C
Post Time: 5 minutes
Injection vol.: 1µL
Flow rate: 0.4mL / minute
Detection: UV Diode Array
Notes: Among the most important purine nucleotides, XMP and GTP are often analyzed from the lymphocytes of healthy people and HIV-1 seropositive patients at different stages of the disease (ARC-AIDS). Several differences in metabolism of Purine Nucleotides in the lymphocytes of the AIDS patients, were observed [2]. XMP does not normally appear in free nucleotide cell extracts, however it is the product of the important cell differentiation Enzyme IMP Dehydrogenase in Ribavirin Therapies.
[1]. “Aqueous normal phase retention of nucleotides on silica hydride- based columns: Method development strategies for analytes relevant in clinical analysis”, M.T. Matyska, J.J. Pesek, J. Duley, M. Zamzami, S.M. Fisher, J. Sep. Sci. (2010/, 33, 930-938.
[2]. “Analysis of purine nucleotides in lymphocytes from healthy subjects and AIDS patients”, A Tabucchi, F Carlucci, E Ramazzotti, MC Re, F Marinello, M Rubino, R Pagani , Biomedecine & Pharmacotherapy, Volume 46, Issue 1, 1992, Pages 25-29.

Attachment
No 113 Purine Nucleotides Analyzed with HPLC pdf 0.2 Mb Download File
This Method achieves the separation of Nucleotides XMP from GTP. The analysis of these polar compounds is achieved at high concentration of an organic Solvent as part of the Mobile Phase which provides increased Sensitivity. Retention times were very Reproducible with %RSD approximately 0.4, even when Red Blood Cell extracts were injected in between the standard samples.



Peaks:
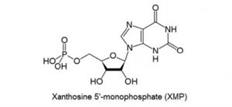
1. XMP: Xanthosine -5’-Monophopate
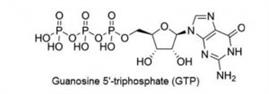
2. GTP: Guanosine -5’-Triphosphate
Column: Cogent Diamond Hydride™, 4µm, 100Å
Catalog No.: 70000-15P-2
Dimensions: 2.1 x 150mm
Mobile Phase:
--A: DI Water + 0.1% Ammonium Formate
--B: 90% Acetonitrile / 10% DI Water / 0.1% Ammonium Formate
Gradient:
| Time (minutes) | %B |
| 0 | 95 |
| 0.5 | 95 |
| 10 | 75 |
| 15 | 30 |
| 20 | 30 |
| 20.1 | 95 |
Post Time: 5 minutes
Injection vol.: 1µL
Flow rate: 0.4mL / minute
Detection: UV Diode Array
Notes: Among the most important purine nucleotides, XMP and GTP are often analyzed from the lymphocytes of healthy people and HIV-1 seropositive patients at different stages of the disease (ARC-AIDS). Several differences in metabolism of Purine Nucleotides in the lymphocytes of the AIDS patients, were observed [2]. XMP does not normally appear in free nucleotide cell extracts, however it is the product of the important cell differentiation Enzyme IMP Dehydrogenase in Ribavirin Therapies.
[1]. “Aqueous normal phase retention of nucleotides on silica hydride- based columns: Method development strategies for analytes relevant in clinical analysis”, M.T. Matyska, J.J. Pesek, J. Duley, M. Zamzami, S.M. Fisher, J. Sep. Sci. (2010/, 33, 930-938.
[2]. “Analysis of purine nucleotides in lymphocytes from healthy subjects and AIDS patients”, A Tabucchi, F Carlucci, E Ramazzotti, MC Re, F Marinello, M Rubino, R Pagani , Biomedecine & Pharmacotherapy, Volume 46, Issue 1, 1992, Pages 25-29.

Attachment
No 113 Purine Nucleotides Analyzed with HPLC pdf 0.2 Mb Download File
