Analytical instrument qualification kits are used to verify performance, support regulatory compliance, and document system suitability for chromatography and spectroscopy instruments. This category includes DIY qualification and performance verification kits for HPLC, UHPLC, and UV‑Vis spectrophotometers, designed for laboratories operating under GxP and regulated quality systems.
These kits provide laboratories with a practical, validated pathway to perform Instrument Qualification (IQ), Operational Qualification (OQ), Performance Qualification (PQ), and HoX (Health‑of‑the‑System) checks in‑house, on their own schedule.
Designed for Regulated Laboratory Environments
Qualification kits in this category are supplied under the Chemical Solutions™ brand and are manufactured using NIST‑traceable reference materials and certified solutions. They are intended to support:
Pharmaceutical and biopharmaceutical laboratories
Contract research organizations (CROs)
Quality control and release labs
Research and analytical development environments
These kits are used to establish and maintain documented instrument performance in accordance with internal SOPs and external regulatory expectations.
Why In‑House Qualification Matters
Instrument qualification is a critical component of compliance programs, method validation, and ongoing data integrity. Performing qualification internally allows laboratories to:
Maintain control over scheduling and documentation
Reduce reliance on external service providers
Perform routine system checks and HoX verification as needed
Rapidly respond to audits, inspections, and investigations
DIY qualification kits provide a structured, repeatable approach to instrument verification while aligning with recognized validation principles.
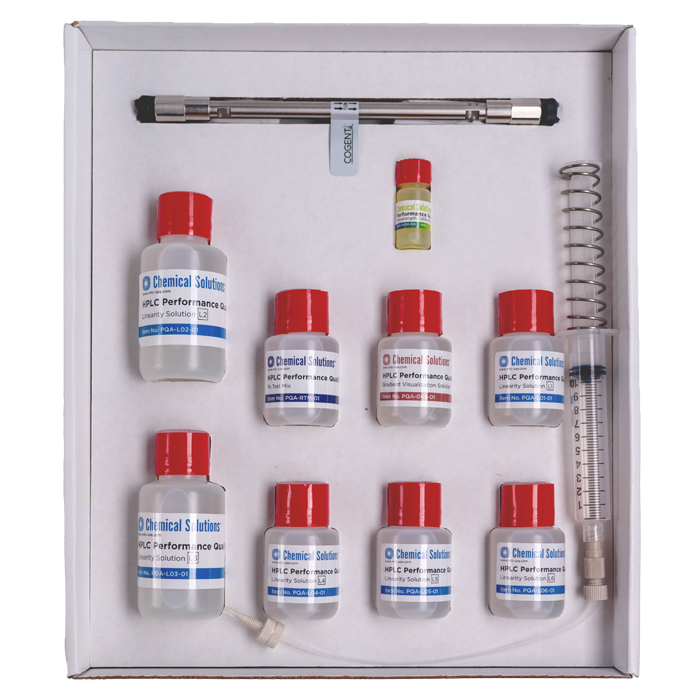
HPLC & UHPLC Qualification Kits (PQ™ & HSQ™)
Chemical Solutions™ PQ™ and HSQ™ kits for HPLC and UHPLC contain all materials required to perform routine performance qualification or performance verification of liquid chromatography systems.
These kits are used to verify:
Precision and repeatability
Accuracy and system response
Instrument performance stability over time
The HSQ™ (High‑Speed Qualification) Kit is designed for faster execution and includes a shorter, high‑efficiency column, enabling rapid qualification while producing equivalent verification results compared to standard PQ™ kits.
Both kits support:
Routine PQ testing
HoX (Health‑of‑Instrument) checks
Ongoing system performance monitoring
UV‑Vis Spectrophotometer Qualification Kits
PQ™ kits for UV‑Vis spectrophotometers provide laboratories with the materials and protocols needed to verify:
Wavelength accuracy
Photometric accuracy
Instrument precision and performance consistency
These kits are commonly used for routine performance verification, validation support, and compliance documentation in quality‑focused laboratory environments.
Qualification Scope & Flexibility
Qualification kits in this category support:
Initial instrument qualification workflows
Routine performance verification
Internal SOP‑driven compliance programs
Audit and inspection readiness
Because the kits are designed for in‑house use, laboratories can perform qualification:
On demand
At defined intervals
After maintenance or system changes
This flexibility supports continuous compliance rather than reactive validation.
Applications & Use Cases
Qualification kits for HPLC and UV‑Vis are commonly used for:
IQ/OQ/PQ programs
HoX and system health monitoring
Method validation and transfer
Audit preparation and compliance maintenance
Pharmaceutical, bioanalytical, and regulated testing environments
Why Choose Chemical Solutions™ Qualification Kits
NIST‑traceable reference materials
Designed for DIY, in‑house qualification
Supports recurring compliance workflows
Compatible with standard laboratory SOPs
Enables cost‑effective, scheduled instrument verification
Related Categories